In the product packaging development process, delays rarely come from a single technical failure. More often, they happen when teams move forward with different assumptions. R&D defines how the product should perform. Engineers focus on how to build it. Each function is valid, but during product packaging prototyping, these perspectives collide.
A dispensing system may not match formulation viscosity. Heat or light elements may affect ingredient stability. Applicator precision may not align with real usage. These issues do not appear at the concept stage. They surface later, during testing, when changes are more expensive and timelines are already tight. This is why packaging and applicator development is not just a design task. It is a coordination problem.
The product packaging development process is the structured design and engineering of packaging systems and applicators that deliver a cosmetic formulation effectively. It includes concept definition, engineering design, product packaging prototyping, validation, and preparation for manufacturing.
Unlike traditional packaging, modern skincare applicator development must account for how the formula behaves during use. Packaging is no longer a passive container. It functions as a delivery system that directly influences performance, consistency, and user experience.
During the engineering design process, three perspectives must align:
Nuon does not develop formulations, but packaging must still be engineered around how the formula behaves.
Without structured cross-functional collaboration, common issues appear during product packaging prototyping:
These are not engineering failures. They are alignment failures..
When alignment happens early, packaging systems are designed with real formulation behavior in mind. For example, viscosity directly impacts flow rate, dosing accuracy, and applicator geometry. If this is not considered upfront, prototypes often require redesign after testing. By integrating formulation insights early, teams reduce late-stage conflicts and shorten iteration cycles without compromising performance.
The engineering design process becomes more focused when requirements are clearly defined before development starts.
Instead of reacting to issues during testing, engineers can:
This reduces unnecessary revisions and improves development speed across the new product packaging development process.
A prototype is not the end goal. It must transition into manufacturing while maintaining performance and compliance.
Early alignment ensures:
This reduces the risk of redesign during scale-up and supports a smoother path to commercialization.
In both medical device and cosmetic engineering workflows, early-stage alignment is widely recognized as a key factor in reducing development risk.
Studies on concurrent engineering and cross-functional collaboration show that integrating multiple disciplines early can significantly reduce redesign cycles and improve product quality.
However, results depend on execution. Alignment must be structured, not informal. Without a clear framework, teams still default to siloed decision-making, and the same issues reappear during prototyping.
At Nuon Medical, packaging and applicator development is approached as a coordinated system, not a sequence of isolated steps.
Through the C.U.R.V.E framework, key variables are aligned early:
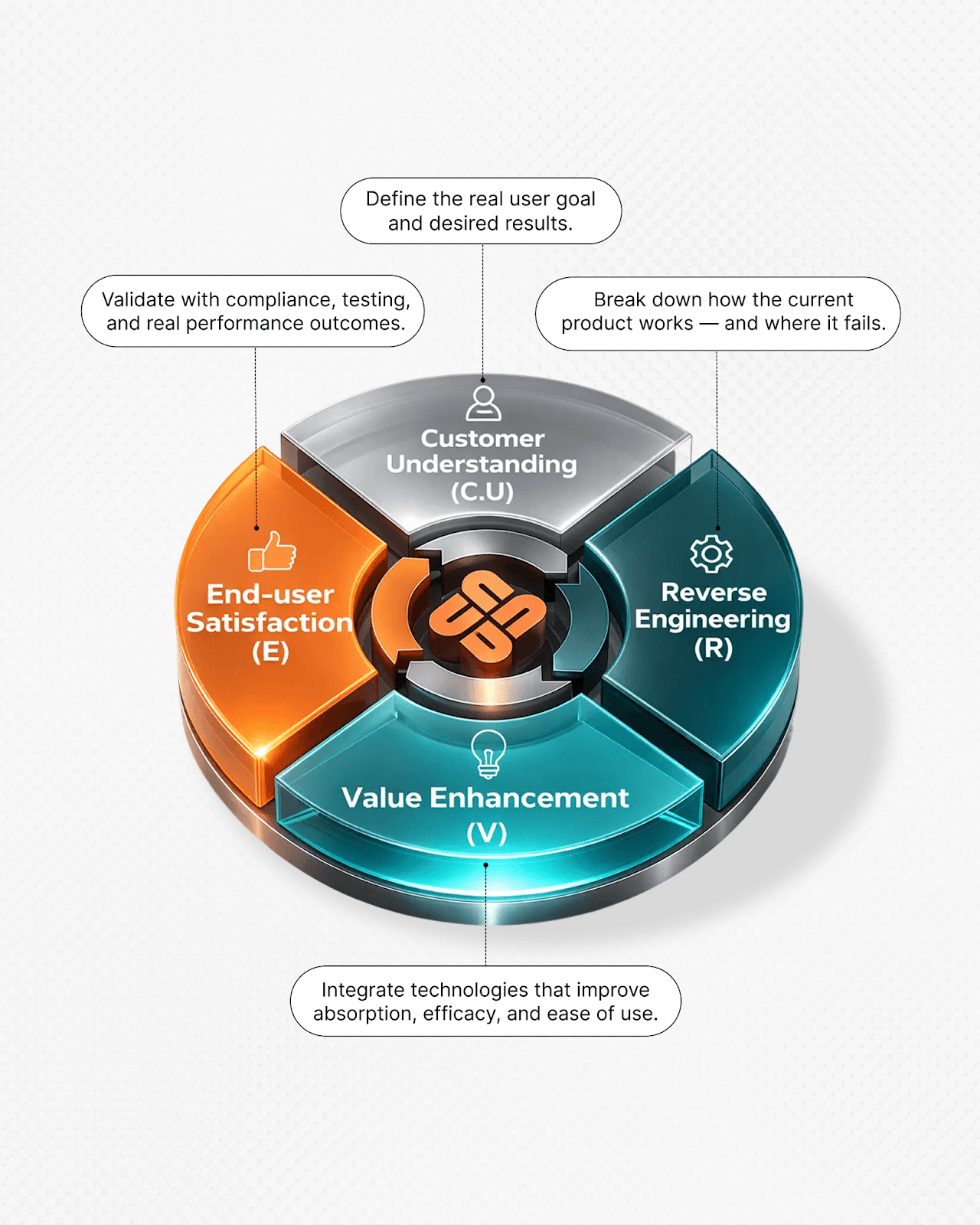
This approach reflects how Nuon engineers package around formulation behavior without modifying the formulation itself.
The result is a product packaging prototyping process that is more predictable, with fewer conflicts and stronger commercial readiness.
1. What is the product packaging development process?
It is the structured process of designing, prototyping, and validating packaging and applicators to ensure they deliver the formulation effectively, consistently, and at scale.
2. Why is cross-functional collaboration important in packaging prototyping?
Because packaging performance depends on multiple variables. Aligning R&D, engineering, and formulation insights early helps prevent technical conflicts and reduces redesign cycles.
3. How does packaging influence product performance?
Packaging and applicators control dosing, application method, and interaction with the skin. Well-designed systems can improve consistency, user experience, and perceived efficacy without changing the formula.
Explore how structured packaging and applicator development can support more efficient, scalable product launches.